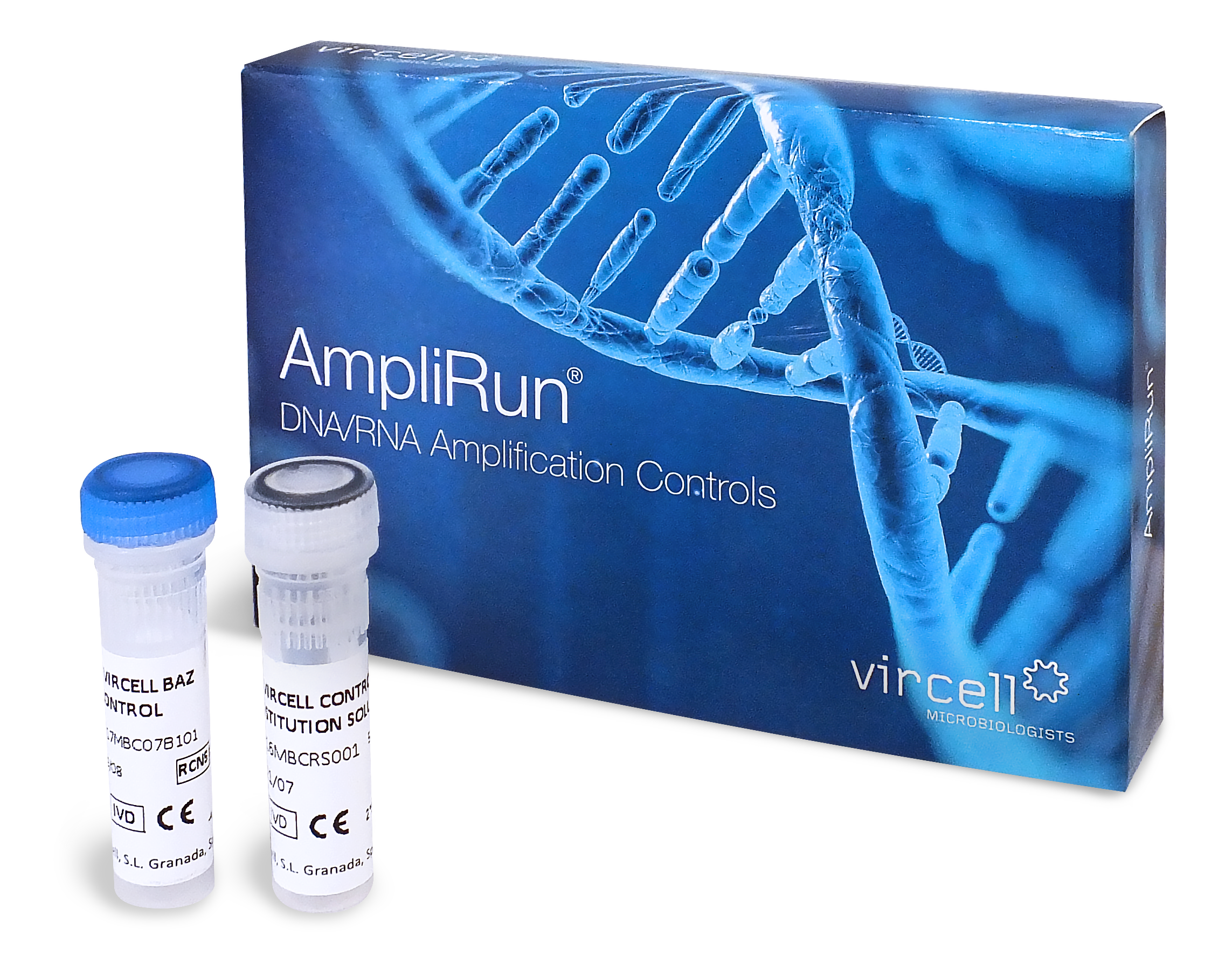
Purified DNA of Borrelia to be used to control research techniques based in nucleic acids amplification.
- Purified complete microbial genome
- Any sequence can be amplified
- Concentration range: 10.000-20.000 copies/µl determined by qPCR
- Can be used in any molecular testing platform
- Non infectious
- Lyophilized presentation
- A resuspension vial is provided within the kit with Molecular Biology Grade Water
> Further information available for the design of primers and probes:
· Complete genome
Borrelia afzelii: CP160066
Borrelia burgdorferi: CP158509
Borrelia garinii CP160381
Ensure accuracy and reproducibility of your molecular tests for infectious diseases with Vircell’s quantified PCR controls.
Take advantage of its wide use:
- Amplification of the genotype of interest
- Validation of in-house and commercial methods (sensitivity, specificity, limit of detection, precision)
- Absolute sample quantification by standard curve
- Objective interpretation of results
- Overcome limitations of using clinical samples as positive controls
Lyophilized presentation improves stability and avoids extra transport costs as there is no need of dry ice. Controls must be reconstituted with 100 µl of Molecular Biology Grade Water, free of DNase, RNase.
Ensure accuracy and reproducibility of your molecular tests for infectious diseases with Vircell’s quantified PCR controls.
Take advantage of its wide use:
- Amplification of the genotype of interest
- Validation of in-house and commercial methods (sensitivity, specificity, limit of detection, precision)
- Absolute sample quantification by standard curve
- Objective interpretation of results
- Overcome limitations of using clinical samples as positive controls
Lyophilized presentation improves stability and avoids extra transport costs as there is no need of dry ice. Controls must be reconstituted with 100 µl of Molecular Biology Grade Water, free of DNase, RNase.
